The configuration of these electrons follows from the principles of quantum mechanics. Since the number of electrons and their arrangement are responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Each electron is influenced by the electric fields produced by the positive nuclear charge and the other (Z – 1) negative electrons in the atom. Therefore, the number of electrons in neutral atom of Magnesium is 12. The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. Magnesium-26 is composed of 12 protons, 12 neutrons, and 12 electrons. Magnesium-25 is composed of 12 protons, 13 neutrons, and 12 electrons. Magnesium-24 is composed of 12 protons, 12 neutrons, and 12 electrons. The lighter isotopes mostly decay to isotopes of sodium while the heavier isotopes decay to isotopes of aluminium. The longest-lived radioisotope is 28Mg with a half-life of 20.915 hours. Magnesium naturally occurs in three stable isotopes, 24Mg, 25Mg, and 26Mg. Mass numbers of typical isotopes of Magnesium are 24 25 26. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The name "Magnesium" originates from a Greek district in Thessaly called Magnesia.Magnesium is a chemical element with atomic number 12 which means there are 12 protons in its nucleus. Commercially, magnesium is primarily used in the creation of strong and lightweight aluminum-magnesium alloys, which have numerous advantages in industrial applications. 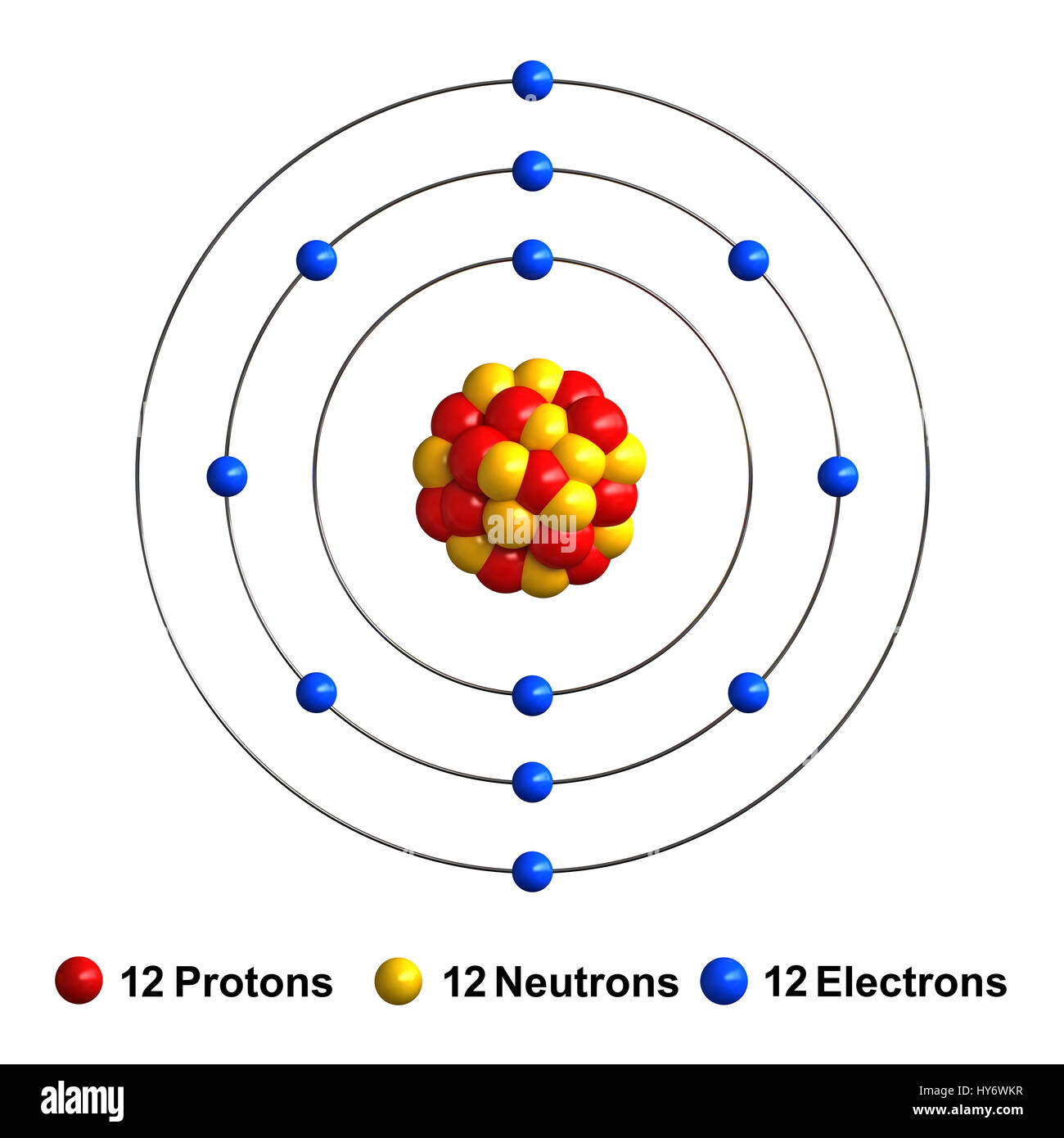
It is can be found in minerals such as brucite, carnallite, dolomite, magnesite, olivine and talc. In its elemental form, magnesium has a shiny grey metallic appearance and is an extremely reactive. Magnesium is the eighth most abundant element in the earth's crust and the fourth most common element in the earth as a whole. Magnesium was discovered by Joseph Black in 1775 and first isolated by Sir Humphrey Davy in 1808. The magnesium atom has a radius of 160 pm and a Van der Waals radius of 173 pm. The number of electrons in each of Magnesium's shells is and its electron configuration is 3s 2. Magnesium (atomic symbol: Mg, atomic number: 12) is a Block S, Group 2, Period 3 element with an atomic mass of 24.3050. Additional technical, research and safety (MSDS) information is available as is a Reference Calculator for converting relevant units of measurement. Typical and custom packaging is available. American Elements produces to many standard grades when applicable, including Mil Spec (military grade) ACS, Reagent and Technical Grade Food, Agricultural and Pharmaceutical Grade Optical Grade, USP and EP/BP (European Pharmacopoeia/British Pharmacopoeia) and follows applicable ASTM testing standards. Magnesium Metal 25 isotopic material is generally immediately available. For thin film applications it is available as rod, pellets, pieces, granules and sputtering targets and as either an ingot or powder. Magnesium Metal is also available in ultra high purity and as nanoparticles. Magnesium 25 Metal is one of over 250 stable Metallic isotopes produced by American Elements for biological and biomedical labeling, as target materials and other applications. It is both naturally occurring and a produced by fission. Magnesium 25 Metal (Magnesium-25) is a stable (non-radioactive) isotope of Magnesium. Thin Film Deposition & Evaporation Materials.Additive Manufacturing & 3D Printing Materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
